This article was originally published in the March 2018 issue of the Journal of Critical Psychology, Counselling and Psychotherapy: Sedarati, J. (2018). Do children diagnosed with ‘ADHD’ have a brain disorder? The Journal of Critical Psychology, Counselling and Psychotherapy, 18(1), 19-29.
SUMMARY: Contemporary literature is reviewed to determine whether there exists evidence supporting the notion that children diagnosed with ‘ADHD’ have a brain disorder.
KEYWORDS: attention-deficit hyperactivity disorder, ADHD, brain disorder
‘Attention-deficit/hyperactivity disorder’ is portrayed as a neurodevelopmental disorder (American Psychiatric Association, 2013), even though ‘no biological marker is diagnostic for ADHD’ (p. 61). The subjective (e.g, ‘often talks excessively’; p. 60) and unreliable (Kirk, Gomory, & Cohen, 2013; Reid & Maag, 1994) criteria used to diagnose ‘ADHD’ has led critics to conclude that it is not a valid medical syndrome (Breggin, 2013) but rather, ‘a grouping of socially disapproved behaviors falsely passed off as a disease’ (Joseph, 2006, p. 246). Despite these criticisms, stimulants are being prescribed to millions of diagnosed children (Zuvekas & Vitiello, 2012), inpart, based on the belief that they have a brain disorder (Timimi, 2009).
Neuroimaging technology cannot aid in the diagnosis of ‘ADHD’ (Castellanos et al., 2002), and has many limitations (Satel & Lilienfeld, 2013; Reeves, Mills, Billick, & Brodie, 2003; Davatzikos, 2004). Moreover, neuroimaging research appears to have provided little justification for framing ‘ADHD’ as a brain disorder. For example, Baumeister and Hawkins’ (2001) review concluded that ‘the neuroimaging literature provides no convincing evidence for the existence of abnormality in the brains of persons with ADHD’ (p. 8). Subsequently, it was reported that most studies (e.g., Castellanos et al., 1994; Filipek et al., 1997) fail to control for the effects of stimulants (Leo & Cohen, 2003), which have been shown to alter the brain (Breggin, 2013). Cohen and Leo (2004) suggested that to determine the effect of stimulants on brain volume, researchers need simply compare medicated ‘ADHD’ children to a control group of age- and weight-matched medication-naïve ‘ADHD’ children. Bush and colleagues (2005) noted that many other factors (e.g., caffeine use) are also ‘under-studied and often ignored’ (p. 1282), however, they neglected to mention trauma; which is associated with ‘ADHD’ (Ford et al., 2000; Kessler et al., 2010; Read & Mayne, 2017) and can alter the brain (Read, Fosse, Moskowitz, & Perry, 2014). Despite these issues, researchers have reported that ‘ADHD’ is associated with enlarged hippocampi (Plessen et al., 2006), and volume reductions in numerous other brain regions including the prefrontal cortex, anterior cingulate (Bush, Valera, & Seidman, 2005), caudate, putamen, globus pallidus (Nakao, Radua, Rubia, & Mataix-Cols, 2011), and cerebellum (Valera, Faraone, Murray, & Seidman, 2007). Although, Ioannidis (2011) has exposed considerable bias in the psychiatric neuroimaging literature.
The present review aimed to provide an updated, critical analysis of the ‘ADHD’ neuroimaging literature, to determine whether the limitations of prior studies (see Leo & Cohen, 2003) remain; or if as suggested by Cohen and Leo (2004), researchers have finally conducted an ‘ADHD’ neuroimaging study which controls for the confounding effects of stimulant drugs. The review’s main conclusion is that defining ‘ADHD’ as a brain disorder remains unsupported by the evidence.
Review methods
Eligible studies were ‘ADHD’ MRI studies published since the update (Cohen & Leo, 2004) to Leo and Cohen’s (2003) review. Studies needed to include a control group of age- and weight-matched medication-naïve ‘ADHD’ children, diagnosed using DSM-IV, DSM-IV-TR, or DSM-5 (APA, 1994, 2000, 2013) criteria. Studies that supplied detailed medication histories, included a trauma survey (e.g., ACE questionnaire; Felitti et al., 1998), and declared no conflicts of interest, were also preferred.
Review findings
An extensive search of electronic databases (PsychINFO, PsychARTICLES, and Google Scholar) was conducted between April and June 2017 using the following keywords: attention deficit hyperactivity disorder, ADHD, brain differences, MRI, brain abnormalities, unmedicated, and medication naïve. Forty articles were analysed for relevance, with the five studies that most closely mirrored the eligibility criteria included in the final review (see Table 1 for key details).
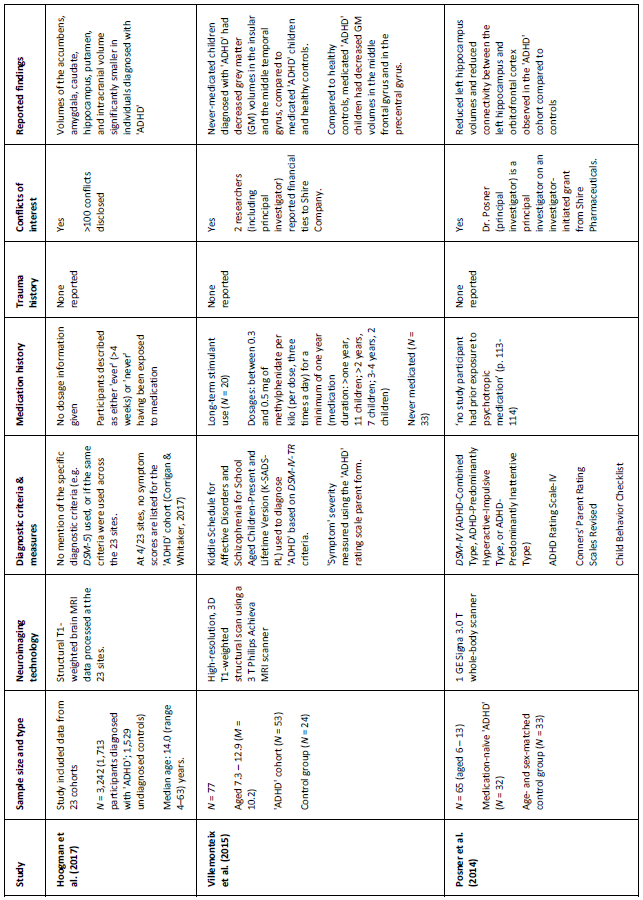
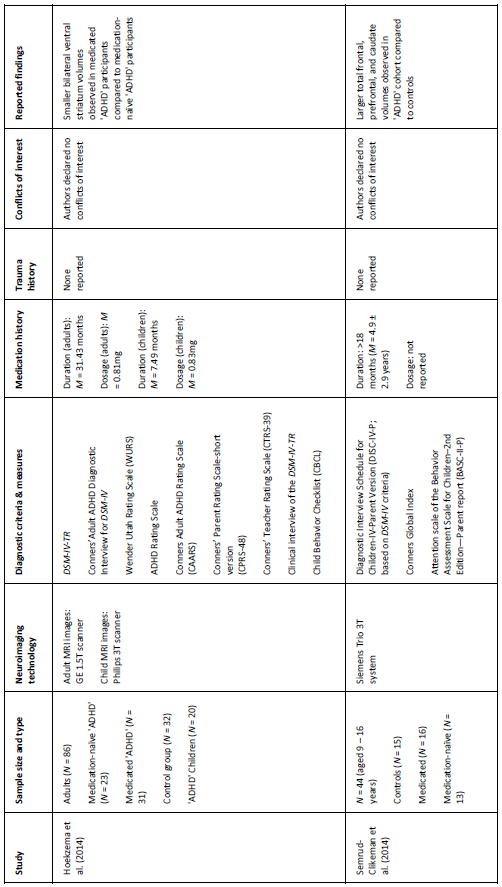
Table 1. Key details of five ‘ADHD’ MRI studies (see appendix for additional information)
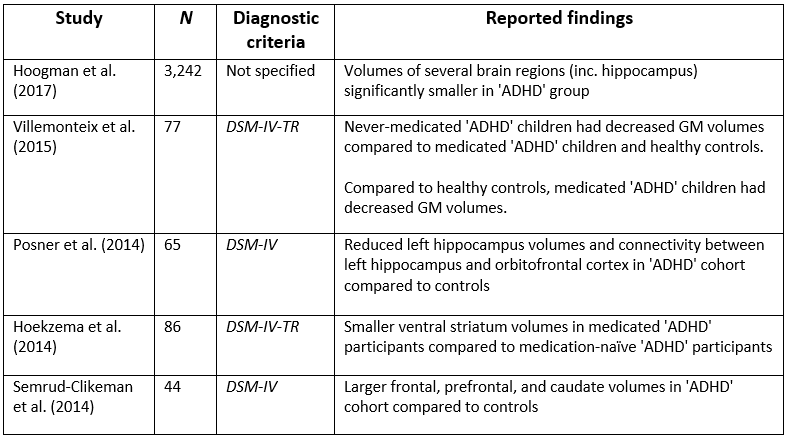
The largest neuroimaging study of ‘ADHD,’ to date (Hoogman et al., 2017), used data (N = 3,242) from the international ENIGMA Working Group collaboration to conduct a cross-sectional ‘mega-analysis’ comparing MRI brain scans of individuals (age range: 4–63 years) diagnosed with ‘ADHD’ with a group of undiagnosed controls. The study reported reduced volumes of the accumbens, amygdala, caudate, hippocampus, putamen, and intracranial volume, in the ‘ADHD’ cohort. However, effect sizes were small (d = -0.10 to -0.19), meaning that the mean brain volumes of the ‘ADHD’ and control group’s largely overlapped. Also, only hippocampus volumes were significantly different in adolescents (15–21 years) and no significant differences were observed in adults (>21 years). Despite these small effects, the researchers concluded: ‘patients with ADHD have altered brains; therefore ADHD is a disorder of the brain’ (p. 2). This is a faulty conclusion because one cannot infer that individuals have disordered brains based merely on mean differences in pooled brain volumes (Corrigan & Whitaker, 2017).
Contrary to Hoogman and colleagues (2017), who observed smaller hippocampi in their ‘ADHD’ cohort, Posner et al., (2014) found that children diagnosed with ‘ADHD’ (N = 32) had enlarged left hippocampi compared to control participants (N = 33). This disparity may be due to the fact that Posner et al. studied medication-naïve participants, exclusively. However, an earlier study of both medicated and medication-naïve participants also reported enlarged hippocampal volumes in children and adolescents (Plessen et al., 2006).
Villemonteix and colleagues’ (2015) cross-sectional study obtained MRI brain scans (N = 77) from both medicated and medication-naïve children diagnosed with ‘ADHD,’ and compared them to an undiagnosed control group. The researchers reported that medication-naïve children had decreased grey matter (GM) volumes in the insular and the middle temporal gyrus, compared to the medicated and control cohorts. Additionally, comparisons between the controls and medicated children showed that the latter group had decreased GM volumes in the middle frontal gyrus and in the precentral gyrus.
Semrud-Clikeman et al. (2014) compared MRI brain scans from three groups (medication-naïve ‘ADHD,’ chronically treated ‘ADHD,’ and control participants) and observed larger prefrontal regions in the ‘ADHD’ cohorts, regardless of medication history, compared to controls. They also found that medication-naïve children had smaller anterior cingulate volumes compared with other groups. This study is limited by its small sample size (N = 44), although previous studies (see Bush et al., 2005) have also suggested that these brain regions contribute to ‘ADHD.’
Hoekzema et al (2014) compared the brains (N = 86) of medication-naïve ‘ADHD’ adults, medicated ‘ADHD’ adults, and a group of control participants, obtained over a 4-year period. Additionally, they acquired scans before and after exposure to methylphenidate in a group of medication-naïve adults and children, diagnosed with ‘ADHD.’ Medicated ‘ADHD’ participants (both children and adults) were found to have smaller bilateral striatum volumes, compared to the medication-naïve cohorts. The researchers suggested that this difference may be due to stimulant-exposure rather than being a feature of ‘ADHD.’ This is a plausible conclusion given the brain changes that can occur whilst consuming stimulants (Breggin, 2013).
Medication
Hoogman et al. (2017) categorised participants as either ‘ever’ (>4 weeks) or ‘never’ having consumed medication. This is problematic because the lasting effects of stimulants may vary widely based on ‘treatment’ duration (see Swanson et al., 2017; Breggin, 2013). Villemonteix et al. (2015) and Hoekzema et al. (2014) provided more detailed medication histories (see appendix), including both duration and dosage estimates; whereas Semrud-Clikeman et al. (2014) provided duration but not dosage estimates. All of the participants in Posner and colleagues’ (2014) study were medication-naïve. Hoogman et al., claimed that the brain differences they observed were not caused by medication effects but neglected to report the proportion of adults versus children who were medication-naïve versus medication-exposed. This is a major confound that undermines the authors’ claim; especially given the fact that most of the statistically significant differences were unique to children (<15 years; Corrigan & Whitaker, 2017).
Conflicts of interest
Researchers from three out of five studies (Hoogman et al., 2017; Villemonteix et al., 2015; Posner et al., 2014) reported conflicts of interest; including the principal investigators, Villemonteix and Posner. These conflicts may have influenced study outcomes (Ahn et al., 2017; also see Whitaker & Cosgrove, 2015). Most notably, Hoogman and colleagues declared over one hundred conflicts (p. 9), including speaking fees from pharmaceutical companies, a patent for an ‘ADHD’ drug treatment, and royalties from a copyrighted rating scale used to diagnose ‘ADHD.’ Clearly the researchers stand to gain from the continued diagnosis and ‘treatment’ of ‘ADHD.’
Additional limitations
The studies under review suffer from many other limitations, only a few of which, are mentioned here. Firstly, in Hoogman and colleagues’ (2017) study, the diagnostic criteria were not standardised across the 23 scan sites. Therefore, different sites may have used different classification methods (e.g., DSM-IV, ICD-10) to diagnose ‘ADHD.’ Secondly, none of the studies conducted a survey to control for the potentially brain-altering effects of trauma (Read et al., 2014). Thirdly, none of the studies controlled for participant body-size. Finally, none of the studies reported using a random sampling method – thus, their findings are difficult to generalise.
Inconsistencies and faulty assumptions
The reviewed literature remains hampered by myriad limitations, inconsistencies, and faulty assumptions, which do not justify defining ‘ADHD’ as a brain disorder. Firstly, all five studies assumed that ‘ADHD’ is a valid medical diagnosis that can reliably classify individuals. This is untrue (Kirk et al., 2013; Reid & Maag, 1994). Neuroimaging technology cannot distinguish ‘ADHD children’ from ‘typically developing children’ (Castellanos et al., 2002), and the use of the DSM’s subjective criteria render all research in this field inherently confounded. Secondly, although all but one (Hoogman et al., 2017) of the studies directly compared age-matched controls to medication-exposed and/or medication-naïve ‘ADHD’ participants, none controlled for body size. Therefore, Leo and Cohen’s (2003) request for researchers to simply compare age- and weight-matched medicated and medication-naïve ‘ADHD’ participants appears to remain unmet. Thirdly, three out of five studies (Posner et al., 2015; Villemonteix et al., 2015; Hoogman et al.) disclosed conflicts of interest, which may have influenced study outcomes (Ahn et al., 2017). Of the two studies without conflicts, one suggested that stimulants may have caused the observed brain differences (Hoekzema et al., 2014), whilst the other suggested that the differences were not causally related to medication-exposure (Semrud-Clikeman et al., 2014). Even if differences in brain volumes are ‘innate’ rather than due to stimulant-exposure, there is still no reason to conclude that they represent a brain disorder (Reeves et al., 2003). Functional and structural differences in skilled individuals (e.g., pianists; Rubia, 2002, in Leo & Cohen, 2003) and victims of child abuse (Read et al., 2014), have demonstrated the brain’s neuroplasticity. Thus, it is not surprising to find differences in brain volumes based on what amounts to differences in behaviour and experience. Moreover, given the absence of recorded trauma histories in the reviewed studies, it’s impossible to say whether the purported brain differences are the result of natural variation or adversity (e.g., maltreatment). This is especially pertinent, given that trauma is associated with ‘ADHD’ (Ford et al., 2000; Kessler et al., 2010; Read & Mayne, 2017).
Although this review was limited by its modest scope, it has demonstrated fundamental shortcomings in the contemporary literature whose ‘biologic tunnel vision’ (Baumeister & Hawkins, 2001, p. 8) has continued to produce similarly unconvincing results to those of decades-past (Leo & Cohen, 2003). Future studies should include an adverse experiences scale to control for the potentially brain-altering effects of trauma (Read et al., 2014). However, this would not solve the underlying issue with research of this kind, which is that there is no way to objectively distinguish ‘normal’ and ‘disordered’ individuals (Kirk et al., 2013).
To summarise …
Millions of children are being prescribed stimulant medication, in-part, based on the belief that they have a brain disorder called ‘attention deficit hyperactivity disorder.’ Attempts to locate purported abnormalities in diagnosed children’s brains have been inconsistent and hampered by confounding variables and other limitations. A review of contemporary neuroimaging studies was conducted in order to provide an updated and critical perspective on the status of ‘ADHD’ as a brain disorder. An extensive search of electronic databases (PsychINFO, PsychARTICLES, and Google Scholar) between April and June 2017 culminated in the selection of five publications that most closely mirrored the review’s eligibility criteria. The reviewed MRI studies do not provide convincing evidence that children diagnosed with ‘ADHD’ have a brain disorder. The studies were limited by conflicts of interest and the use of invalid and unreliable diagnostic criteria. They also failed to control for the potentially brain-altering effects of childhood trauma. Future researchers should control for this confounding variable by including an adverse experiences scale in their studies. However, neuroimaging studies of ‘ADHD’ suffer from a more profound issue, which is that researchers are incapable of objectively distinguishing ‘disordered’ and ‘typically developing’ children. Until this issue is addressed, all studies of this kind will remain suspect.
Ahn, R., Woodbridge, A., Abraham, A., Saba, S., Korenstein, D., Madden, E., . . . Keyhani, S. (2017). Financial ties of principal investigators and randomized controlled trial outcomes: cross sectional study. British Medical Journal, 356(i6770), 1-9.
American Psychiatric Association. (1994). Diagnostic and statistical manual of mental disorders (4th ed.). Washington, DC: Author.
American Psychiatric Association. (2000). Diagnostic and statistical manual of mental disorders (4th ed.). Washington, DC: Author.
American Psychiatric Association. (2013). Diagnostic and statistical manual of mental disorders (5th ed.). Washington, DC: Author.
Baumeister, A. A., & Hawkins, M. F. (2001). Incoherence of neuroimaging studies of attention deficit/hyperactivity disorder. Clinical Neuropharmacology, 24(1), 2-10.
Breggin, P. R. (2013). Psychiatric drug withdrawal: A guide for prescribers, therapists, patients, and their families. New York: Springer.
Bush, G., Valera, E. M., & Seidman, L. J. (2005). Functional neuroimaging of attention-deficit/hyperactivity disorder: A review and suggested future directions. Biological Psychiatry, 57, 1273-1284. doi:10.1016/j.biopsych.2005.01.034
Castellanos, F. X., Giedd, J. N., Eckburg, P., Marsh, W. L., Vaituzis, C., Kaysen, D., . . . Rapoport, J. L. (1994). Quantitative morpohology of the caudate nucleus in attention deficit hyperactivity disorder. American Journal of Psychiatry, 151(12), 1791-1796.
Castellanos, F. X., Lee, P. P., Sharp, W., Jeffries, N. O., Greenstein, D. K., Clasen, L. S., . . . Rapoport, J. L. (2002). Developmental trajectories of brain volume abnormalities in children and adolescents with attention-deficit/hyperactivity disorder. Journal of the American Medical Association, 288(14), 1740-1748. doi:10.1001/jama.288.14.1740
Cohen, D., & Leo, J. (2004). An update on ADHD neuroimaging research. The Journal of Mind and Behavior, 25(2), 161-166.
Corrigan, M. W., & Whitaker, R. (2017, April 15). Lancet Psychiatry needs to retract the ADHD-Enigma study. Retrieved June 27, 2017, from Mad in America: https://www.madinamerica.com/2017/04/lancet-psychiatry-needs-to-retract-the-adhd-enigma-study/
Davatzikos, C. (2004). Why voxel-based morpometric analysis should be used with great caution when characterizing group differences. NeuroImage, 23, 17-20. doi:10.1016/j.neuroimage.2004.05.010
Felitti, V., Anda, R. F., Nordenberg, D., Williamson, D. F., Spitz, A. M., Edwards, V., . . . Marks, J. S. (1998). Relationship of childhood abuse and household dysfunction to many of the leading causes of death in adults: The Adverse Childhood Experiences (ACE) study. American Journal of Preventive Medicine, 14(4), 245-258.
Filipek, P. A., Semrud-Clikeman, M., Steingard, R. J., Renshaw, P. F., Kennedy, D. N., & Biederman, J. (1997). Volumetric MRI analysis comparing subjects having attention-deficit hyperactivity disorder with normal controls. Neurology, 48, 589-601.
Ford, J. D., Racusin, R., Ellis, C. G., Reiser, J., Fleischer, A., & Thomas, J. (2000). Child maltreatment, other trauma exposure, and posttraumatic stress symptomatology among children with oppositional defiant disorder and attention deficit hyperactivity disorder. Child Maltreatment, 5(3), 205-217.
Hoekzema, E., Carmona, S., Ramos-Quiroga, J. A., Canals, C., Moreno, A., Fernandez, V. R., . . . Vilarroya, O. (2014). Stimulant drugs trigger transient volumetric changes in the human ventral striatum. Brain Struc. Func., 219, 23-34.
Hoogman, M., Bralten, J., Hibar, D. P., Mennes, M., Zwiers, M. P., Schweren, L. S., & van Hulzen, K. J. (2017). Subcortical brain volume differences in participants with attention deficit hyperactivity disorder in children and adults: a cross-sectional mega-analysis. The Lancet, 4, 310–319.
Ioannidis, J. P. (2011). Excess significance bias in the literature on brain volume abnormalities. Archives of General Psychiatry, 68(8), 773-780. doi:10.1001/archgenpsychiatry.2011.28
Joseph, J. (2006). The missing gene: Psychiatry, heredity, and the fruitless search for genes. New York: Algora.
Kessler, R. C., McLaughlin, K. A., Green, J. G., Gruber, M. J., Sampson, N. A., Zaslavsky, A. M., . . . Benjet, C. (2010). Childhood adversities and adult psychopathology in the WHO World Mental Health Surveys. The British Journal of Psychiatry, 197(5), 378-385. doi:10.1192/bjp.bp.110.080499
Kirk, S. A., Gomory, T., & Cohen, D. (2013). Mad science: psychiatric coercion, diagnosis, and drugs. New Brunswick: Transaction.
Leo, J., & Cohen, D. (2003). Broken brains or flawed studies? A critical review of ADHD neuroimaging research. The Journal of Mind and Behavior, 24(1), 29-56.
Nakao, T., Radua, J., Rubia, K., & Mataix-Cols, D. (2011). Gray matter volume abnormalities in ADHD: voxel-based meta-analysis exploring the effects of age and stimulant medication. Mechanisms of Psychiatric Illness, 168(11), 1154-1163.
Plessen, K. J., Bansal, R., Zhu, H., Whiteman, R., Amat, J., Quackenbush, G. A., . . . Peterson, B. S. (2006). Hippocampus and amygdala morphology in attention-deficit/hyperactivity disorder. Archives of General Psychiatry, 63, 795-807.
Posner, J., Siciliano, F., Wang, Z., Liu, J., Sonuga-Barke, E., & Greenhill, L. (2014). A multimodal MRI study of the hippocampus in medication-naive children with ADHD: What connects ADHD and depression? Psychiatry Research: Neuroimaging, 224, 112-118. doi:10.1016/j.pscychresns.2014.08.006
Read, J., & Mayne, R. (2017). Understanding the long-term effects of childhood adversities: beyond diagnosis and abuse. Journal of Child and Adolescent Trauma. doi:10.1007/s40653-017-0137-0
Read, J., Fosse, R., Moskowitz, A., & Perry, B. (2014). The traumagenenic neurodevelopmental model of psychosis revisited. Neuropsychiatry, 4(1), 65-79.
Reeves, D., Mills, M. J., Billick, S. B., & Brodie, J. D. (2003). Limitations of brain imaging in forensic psychiatry. Journal of American Academic Psychiatry & Law, 31(1), 89-96.
Reid, R., & Maag, J. W. (1994). How many fidgets in a pretty much: A critique of behavior rating scales for identifying students with ADHD. Journal of School Psychology, 32(4), 339-354.
Satel, S., & Lilienfeld, S. O. (2013). Brainwashed: The seductive appeal of mindless neuroscience. New York: Basic Books.
Semrud-Clikeman, M., Pliszka, S. R., Bledsoe, J., & Lancaster, J. (2014). Volumetric MRI differences in treatment naïve and chronically treated adolescents with ADHD-combined type. Journal of Attention Disorders, 18(6), 511-520. doi:10.1177/1087054712443158
Swanson, J. M. (2017). Young adult outcomes in the follow-up of the multimodal treatment study of attention-deficit/hyperactivity disorder: symptom persistence, source, discrepancy, and height suppresion. The Journal of Child Psychology and Psychiatry, 1-16. doi:10.1111/jcpp.12684
Timimi, S. (2009). The use of psycho-pharmaceuticals to control boys’ behaviour: A tale of badly behaving drug companies and doctors. The Arab Journal of Psychiatry, 20(2), 147-160.
Valera, E. M., Faraone, S. V., Murray, K. E., & Seidman, L. J. (in press). Meta-analysis of structural imaging findings in attention-deficit/hyperactivity disorder. Biological Psychiatry, 1-9. doi:10.1016/j.biopsych.2006.06.011
Villemonteix, T., de Brito, S. A., Kavec, M., Balériaux, D., Metens, T., Slama, H., . . . Massat, I. (2015). Grey matter volumes in treatment naïve vs. chronically treated children with attention deficity/hyperactivity disorder: a combined approach. European Neuropsychopharmacology. doi:10.1016/j.euroneuro.2015.04.015
Whitaker, R., & Cosgrove, L. (2015). Psychiatry under the influence: Institutional corruption, social injury, and prescriptions for reform. New York: Palgrave Macmillan.
Zuvekas, S. H., & Vitiello, B. (2012). Stimulant medication use in children: a 12-year perspective. American Journal of Psychiatry, 169, 160-166.
Appendix – Table 2. Extended details of five ‘ADHD’ MRI studies